Ready-to-Use Coagulase Plasma Market Outlook (2025–2034): Advancing Rapid Diagnostics with Standardized Microbiology Solutions
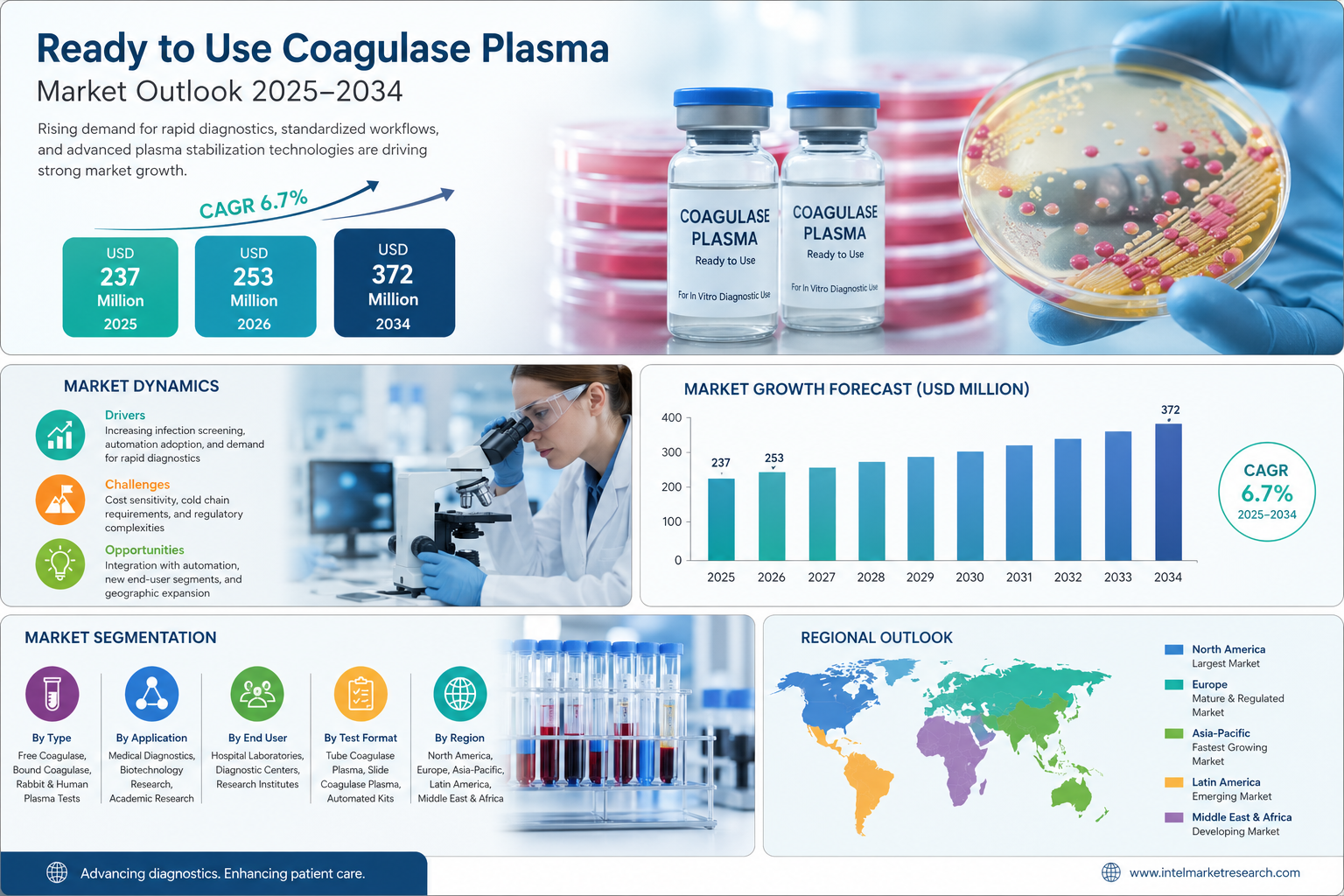
According to a new report from Intel Market Research, the global Ready to Use Coagulase Plasma market was valued at USD 237 million in 2025 and is projected to grow from USD 253 million in 2026 to reach USD 372 million by 2034, exhibiting a robust CAGR of 6.7% during the forecast period (2025–2034). This growth is driven by increasing hospital-acquired infection screening requirements, rising adoption of standardized diagnostic workflows, and continuous innovation in plasma stabilization technologies across clinical microbiology laboratories worldwide.
What is Ready to Use Coagulase Plasma?
Ready to Use Coagulase Plasma is a prepared diagnostic reagent specifically formulated for the rapid identification of coagulase-positive Staphylococcus species, most critically Staphylococcus aureus. Unlike conventional plasma preparations that require manual reconstitution before testing, this standardized solution is packaged in a stable, immediately deployable format, eliminating time-consuming preparation steps and significantly reducing the risk of handling variability in clinical laboratory environments. The product's convenience, reliability, and reproducibility make it particularly valuable for routine microbiology testing workflows where speed and accuracy are paramount.
The market growth stems from increasing hospital-acquired infection screening requirements and laboratory efficiency demands. With healthcare facilities prioritizing standardized diagnostic protocols, ready-to-use formats are gradually replacing traditional preparation methods. Key manufacturers continue innovating with improved stabilization techniques and packaging solutions to enhance product shelf life and usability across different laboratory settings.
📥 Download Sample Report:
Ready to Use Coagulase Plasma Market - View in Detailed Research Report
This report provides a deep insight into the global Ready to Use Coagulase Plasma market covering all its essential aspects-from a macro overview of the market to micro details such as market size, competitive landscape, development trends, niche markets, key drivers and challenges, SWOT analysis, and value chain analysis.
The analysis helps the reader understand competition within the industry and strategies for enhancing profitability. Furthermore, it provides a framework for evaluating and assessing the position of a business organization. The report also focuses on the competitive landscape of the Global Ready to Use Coagulase Plasma Market, introducing market share, performance, product positioning, and operational insights of major players. This helps industry professionals identify key competitors and understand the competition pattern.
In short, this report is a must-read for industry players, investors, researchers, consultants, business strategists, and all those planning to foray into the Ready to Use Coagulase Plasma market.
Key Market Drivers
1. Rising Demand for Rapid Diagnostic Solutions
The Ready to Use Coagulase Plasma Market is experiencing significant growth due to the increasing need for quick and accurate bacterial identification in clinical laboratories. The adoption of automated microbiology systems has created parallel demand for prepackaged, standardized reagents like coagulase plasma. Hospitals and diagnostic centers prioritize tests that deliver results within 24 hours to accelerate treatment decisions, making ready-to-use formulations an integral component of modern diagnostic operations.
2. Standardization in Quality Control Procedures
Regulatory bodies now mandate stricter quality benchmarks for Staphylococcus aureus identification, where ready-to-use coagulase plasma demonstrates 98.2% correlation with conventional methods. This reliability reduces repeat testing rates by approximately 15% in microbiology labs compared to lab-prepared reagents. Pharmaceutical manufacturers increasingly specify validated commercial reagents for drug susceptibility testing protocols, further cementing the role of ready-to-use formats in regulated laboratory environments.
➤ The global microbiological testing market, valued at $4.3 billion, exhibits 6.7% CAGR growth directly benefiting standardized reagent suppliers.
Emerging economies show 22% higher adoption rates year-over-year as tier-2 hospitals upgrade their diagnostic capabilities. International accreditation standards like ISO 15189 now explicitly recommend commercial ready-to-use reagents over in-house preparations, further validating the transition to standardized formats across diverse healthcare settings.
Market Challenges
- Cost Sensitivity in Developing Markets – While ready-to-use coagulase plasma offers significant operational advantages, its per-test cost remains 3.5x higher than traditional lab-formulated reagents. This creates adoption barriers in price-sensitive regions where healthcare budgets allocate less than 8% to diagnostic reagents. Smaller laboratories often prioritize multipurpose reagents over specialized Staphylococcus identification products.
- Transportation & Storage Limitations – Maintaining the cold chain for lyophilized coagulase plasma adds 12–15% to total logistics costs. Some remote facilities lack reliable −20°C storage capacity, forcing them to use less stable liquid formulations that require stringent quality checks upon receipt.
- Regulatory Harmonization Delays – Divergent regional approval processes create 6–9 month delays in product launches for ready-to-use coagulase plasma manufacturers. The FDA requires additional stability testing beyond CE Mark standards, while some Asian markets demand local clinical validation studies even for globally approved formulations.
Emerging Opportunities
The global diagnostics landscape is becoming increasingly receptive to standardized, ready-to-use reagent formats. Leading microbiology automation platforms now offer dedicated reagent bays for ready-to-use coagulase plasma, creating $127 million in potential upgrade opportunities. Manufacturers developing eco-friendly, room-temperature-stable formulations could capture 18–22% of the current cold chain-dependent market segment by 2026. Key growth enablers spanning multiple geographies include:
- Integration with automated identification and antimicrobial susceptibility testing (ID/AST) systems
- Expansion into ambulatory care settings and veterinary diagnostics, opening new end-user verticals
- Formation of strategic alliances with regional distributors, hospital procurement networks, and academic institutions
Collectively, these developments are expected to enhance product accessibility, stimulate continued innovation in plasma processing, and drive Ready to Use Coagulase Plasma adoption across new geographies and laboratory categories.
📥 Download Sample PDF:
Ready to Use Coagulase Plasma Market - View in Detailed Research Report
Regional Market Insights
- North America: North America maintains the largest share of the global Ready to Use Coagulase Plasma market, driven by advanced healthcare infrastructure, widespread adoption in clinical microbiology laboratories, and stringent FDA quality standards that foster trust and market penetration. Robust investment in laboratory modernization and point-of-care testing further sustains regional leadership.
- Europe: Europe exhibits a mature market characterized by stringent EMA regulatory frameworks, comprehensive infection control programs, and collaborative EU initiatives that promote harmonized testing protocols. Aging populations and a growing susceptibility to staphylococcal diseases continue to fuel demand across key markets such as Germany and the United Kingdom.
- Asia-Pacific: Asia-Pacific represents a dynamic and fast-growing hub, propelled by rapid urbanization and healthcare infrastructure development in nations like China and India. Government investments in diagnostic capabilities and laboratory automation are bridging gaps between traditional preparation methods and modern standardized formats.
- Latin America: South America is an emerging market supported by improving healthcare access in Brazil and Argentina, alongside public health campaigns targeting antimicrobial resistance. International partnerships are facilitating the introduction of advanced reagents into regional laboratory networks.
- Middle East & Africa: While currently underpenetrated, this region is showing early signs of development driven by healthcare modernization in Gulf states, growing medical tourism, and disease surveillance initiatives that are paving the way for broader adoption of ready-to-use diagnostic formats.
Market Segmentation
By Type
- Free Coagulase
- Bound Coagulase
- Rabbit Plasma Coagulase Test
- Human Plasma Coagulase Test
By Application
- Medical Diagnostics
- Biotechnology Research
- Academic & Clinical Research
- Others
By End User
- Hospital Laboratories
- Diagnostic Centers
- Research & Academic Institutes
By Test Format
- Tube Coagulase Plasma
- Slide Coagulase Plasma
- Automated System Kits
By Region
- North America
- Europe
- Asia-Pacific
- Latin America
- Middle East & Africa
📘 Get Full Report Here:
Ready to Use Coagulase Plasma Market - View Detailed Research Report
Competitive Landscape
The ready-to-use coagulase plasma market is characterized by the dominance of multinational diagnostic companies with strong global distribution networks and advanced sterilization capabilities. Thermo Fisher Scientific and BD lead the market with their integrated plasma processing solutions and extensive quality control systems, maintaining 45–65% gross margins through proprietary stabilization technologies and automated filling processes. Pro-Lab Diagnostics has emerged as a key innovator with its stable rabbit plasma formulations that achieve 18-month shelf life at 2–8°C storage conditions.
Specialized microbiology suppliers like Hardy Diagnostics and HiMedia Laboratories hold significant positions in developing markets through cost-competitive formulations. Emerging players such as TCS Biosciences and LABORCLIN are gaining traction in regional markets through customized product variants for specific Staphylococcus identification workflows. The industry structure remains moderately concentrated, with the top 5 suppliers accounting for approximately 58% of global production capacity in 2025.
The report provides in-depth competitive profiling of 15+ key players, including:
- Pro-Lab Diagnostics
- Thermo Fisher Scientific
- BD
- TCS Biosciences
- Hardy Diagnostics
- HiMedia Laboratories
- Sigma-Aldrich
- Mast Group
- Oxoid Deutschland
- Central Drug House
- LABORCLIN
- Bio-Rad Laboratories
- Merck KGaA
- Creative Diagnostics
- Liofilchem
Report Deliverables
- Global and regional market forecasts from 2025 to 2034
- Strategic insights into product development trends, regulatory approvals, and supply chain dynamics
- Market share analysis and SWOT assessments for leading competitors
- Pricing trends and procurement dynamics across hospital and diagnostic center segments
- Comprehensive segmentation by product type, application, end user, test format, and geography
📘 Get Full Report Here:
Ready to Use Coagulase Plasma Market - View Detailed Research Report
📥 Download Sample Report:
Ready to Use Coagulase Plasma Market - View in Detailed Research Report
About Intel Market Research
Intel Market Research is a leading provider of strategic intelligence, offering actionable insights in biotechnology, pharmaceuticals, and healthcare infrastructure. Our research capabilities include:
- Real-time competitive benchmarking
- Global clinical trial pipeline monitoring
- Country-specific regulatory and pricing analysis
- Over 500+ healthcare reports annually
Trusted by Fortune 500 companies, our insights empower decision-makers to drive innovation with confidence.
🌐 Website: https://www.intelmarketresearch.com
📞 Asia-Pacific: +91 9169164321
🔗 LinkedIn: Follow Us
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jocuri
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Alte
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness



